Top Bromine Manufacturers in India


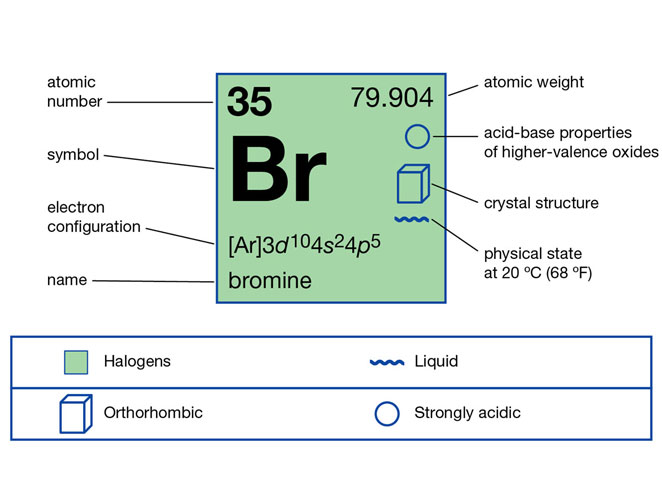



Bromine
Elements are present around us, everywhere, in different forms with different uses and abilities. We use them in any shape, form, product available to make our lives more efficient! But how does that even work? How do elements like Bromine are available for us to use THAT easily?
Bromine is a chemical element which is fuming red liquid at room temperature and its properties are intermediate between those of chlorine and iodine. Bromine in its element is very reactive and thus it is not found in the free state. It is present in colourless salts but due to its high solubility or high affinity to mix in water, it is mostly found in seawater.
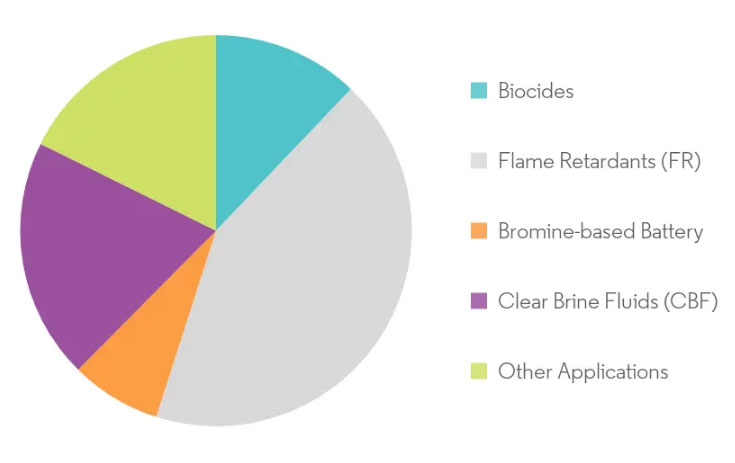
Bromine is widely used in the Agro Chemicals, Pharmaceuticals and fire retardants Products. It is also used in some of the special industrial applications such as Water treatment, Dyes, Chemical of photography, Oil Wells drilling fluids etc.
Jagannath Halogen Pvt Ltd is a part of legacy of almost four decades of experience in the salt industry, being a proud part of Jagannath Salt Group. know more
Our Products
Contact Us
Address:
Survey No. 147/1, 151/1/1, Rann Road, Kharaghoda, Patdi, Surendranagar,
Gujarat - INDIA - 382760 Phone: +91 98871 22262, +91 98292 02631
Email:
Admin - info@jagannathhalogen.com
Sales - crm@jagannathhalogen.com